Congratulations to Hicks lab and Utah CIHD Protein-Metabolite Discovery Core, in collaboration with Kim Orth at UTSW for their recent publication
Inhibition of FicD-mediated AMPylation and deAMPylation by isoprenoid diphosphates
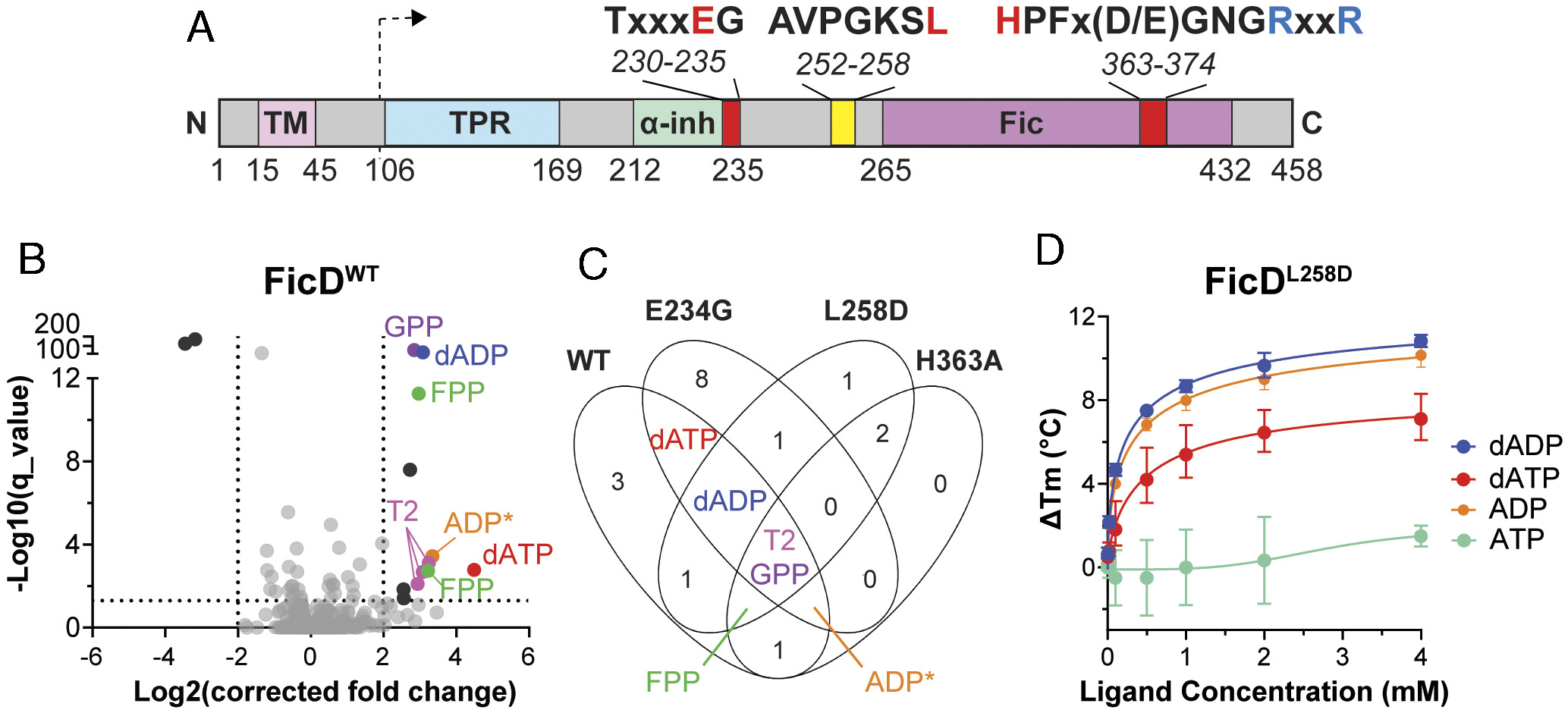
FicD regulates Unfolded Protein Response (UPR) signaling in metazoans by fine-tuning BiP chaperone capacity. Therefore, targeting FicD activity may be a tractable method of altering UPR signaling for therapeutic benefit. We identify geranyl- and farnesyl-pyrophosphate as specific FicD inhibitors. Notably, these small molecules differentially inhibit disease-causing variants of FicD. A structure of farnesyl-pyrophosphate bound to the FicD active site helps explain the differential inhibition of pathogenic variants and provides insight into interactions that can be differentially exploited for modifying FicD activity. Their composition provides a chemical foundation for future drug development efforts targeting FicD activity.